A differentiated treatment for massive irreparable rotator cuff tears (MIRCTs)
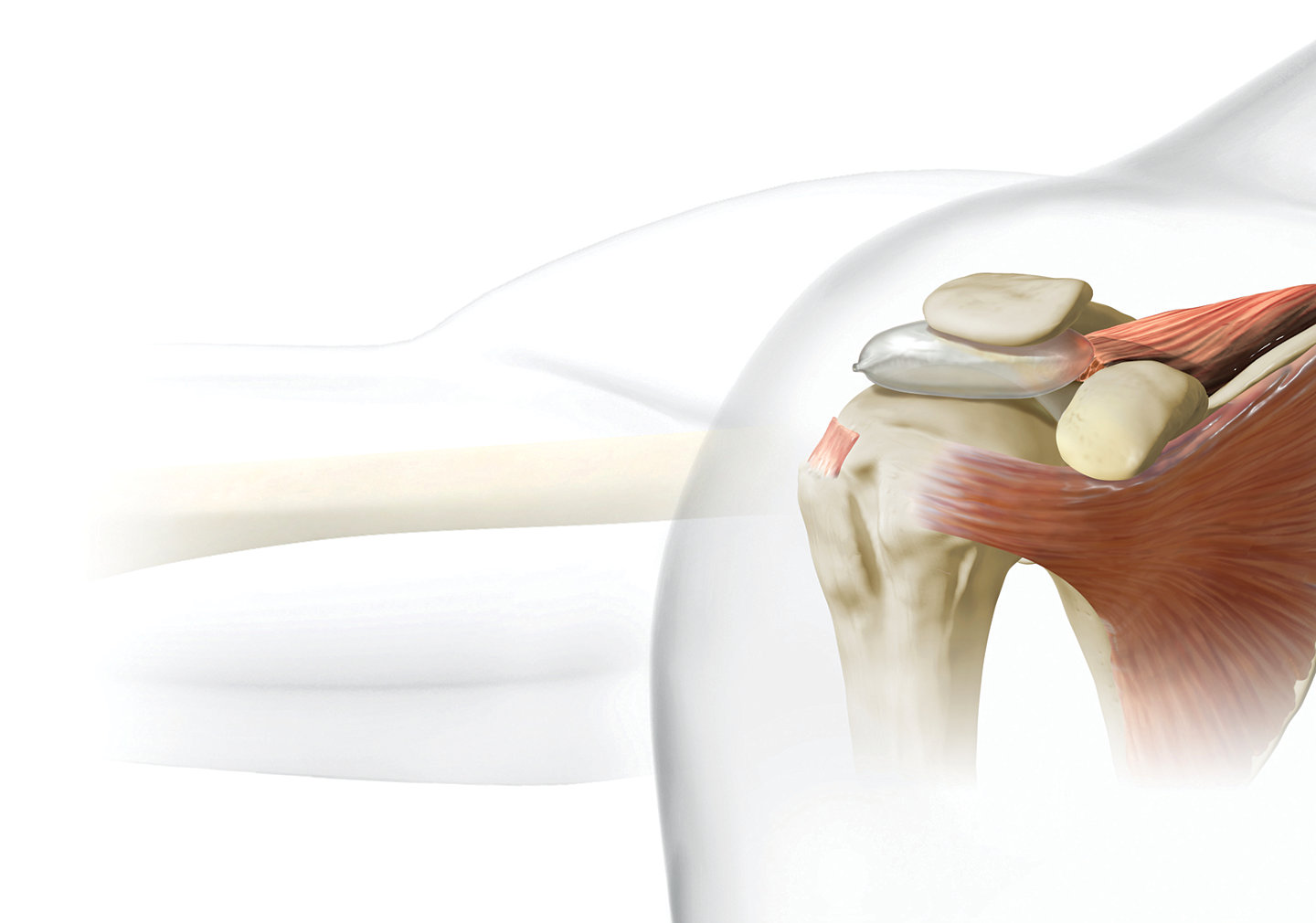
The InSpace subacromial balloon spacer* is designed to restore the subacromial space without sutures or fixation devices and offers patients a minimally invasive, shorter procedure time compared to partial repair. Successful results begin with patient selection. InSpace is recommended for patients1 65+ years old who have:
- Massive, irreparable full thickness rotator cuff tear ≥5cm in diameter
- Involves ≥2 tendons
- Mild – moderate glenohumeral arthritis
- Functional deltoid muscle
- Intact subscapularis
- Intact coracoacromial ligament
- Preserved passive range of motion
- No known neurovascular disease, blood coagulation disorders, or any other condition that would compromise healing
See the efficiency
Watch a short animation of the InSpace procedure demonstrating the deployment and inflation of the balloon in the subacromial space.
InSpace has a long history of changing practice and changing lives across the globe with more than 75,000 patients treated worldwide and 75+ peer-reviewed clinical articles including multiple long-term studies that demonstrated potential sustained benefits at a minimum of 5 years2-3.
InSpace is the industry’s only minimally invasive biodegradable subacromial balloon spacer for MIRCTs backed by level 1 clinical evidence.
The Level 1 Pivotal Study demonstrated1:
Shorter operative time
Early functional recovery
Early pain relief post procedure
Demonstrated safety profile
InSpace fills a gap within the continuum of care for eligible patients with MIRCTs who do not have significant osteoarthritis, have preserved range of motion, and should not undergo a more invasive procedure.

In order of least invasive to most invasive, the shoulder continuum of care can include conservative treatment, debridement, InSpace, partial repair, tendon transfer, SCR, and reverse total shoulder arthroplasty.
Hear from the experts
Watch shoulder specialist, Dr. Ryan Krupp** explain where InSpace fits within his continuum of care.
The American Academy of Orthopaedic Surgeons (AAOS) released a new Evidence-Based Clinical Practice Guideline (CPG) on the Management of Rotator Cuff Injuries. This CPG provides evidence-based recommendations and formally acknowledges balloon spacers as a minimally invasive procedure for irreparable rotator cuff tears that can improve patient-reported outcomes.
Ěý
The InSpace balloon implant helps fill an unmet clinical need for a difficult to treat patient population and presents a streamlined, arthroscopic procedural option that may enable sustained, clinically meaningful improvements for your patients.ĚýContact us today to learn more!Ěý
References
1. Verma N, Srikumaran U, Roden CM, Rogusky EJ, Lapner P, Neill H, Abboud JA. (2022). InSpace implant compared with partial repair for treatment of full-thickness massive rotator cuff tears. J Bone JT Surg Am. Advance online publication. doi. 10.2106/JBJS.21.00667.
2. Kishan A, Russo R, Goldfarb SI, Nelson S, Thomas K, Logoteta M, Giuzio E, Gasparini G, Srikumaran U, Familiari F. Arthroscopic Subacromial Balloon Spacer for Massive Rotator Cuff Tears Demonstrates Improved Shoulder Functionality and High Revision-Free Survival Rates at a Minimum 5-Year Follow-Up. Arthroscopy. 2025 Apr;41(4):886-893. doi: 10.1016/j.arthro.2024.06.024. Epub 2024 Jun 22. PMID: 38914297.
3. Atoun E, Oulianski M, Bachar-Avnieli I, Artamonov A, Gilat R, Lubovsky O, Rosinsky PJ. Subacromial Balloon Spacer for Irreparable Rotator Cuff Tear Treatment Shows Improved Pain and Strength at 5-Year Follow-Up. Arthroscopy. 2024 Feb;40(2):543-550. doi: 10.1016/j.arthro.2023.07.058. Epub 2023 Aug 14. PMID: 37586666.
*InSpace Indications for Use (US): The InSpace subacromial tissue spacer system is indicated for the treatment of patients with massive, irreparable full-thickness torn rotator cuff tendons due to trauma or degradation with mild to moderate gleno-humeral osteoarthritis in patients greater than or equal to 65 years of age whose clinical conditions would benefit from treatment with a shorter surgical time compared to partial rotator cuff repair.
**Stryker consultant
SM-INSP-SYK-2404288

